Topics:
States of Matter:
States of matter, such as solid, liquid, and gaseous, describe how substances behave under different conditions. This is not only relevant in science but also plays a vital role in everyday applications, such as automotive air conditioning. In this article, we delve deeper into what these states signify, particularly in relation to automotive air conditioning. We use the refrigerant R134a as an example to clarify.
Substances can exist in different states: solid, liquid, and gaseous. This depends on how warm or cold it is and the amount of pressure present. At low temperatures, the substance is typically solid, like ice. At higher temperatures, it becomes liquid, like water, and at even higher temperatures, it changes to a gas, such as steam. This change of state is referred to as phase transitions.
Understanding phase transitions is crucial in automotive air conditioning. Here, the refrigerant, such as R134a, is key. The refrigerant continuously changes from liquid to gas and back again. During evaporation, the refrigerant absorbs heat and changes to a gas. It is then compressed, transforming back into a liquid and releasing heat. This repeated cycle ensures the temperature inside the car is properly controlled.
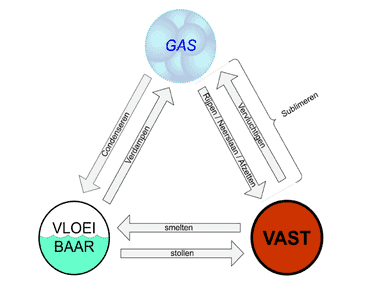
Condensation: Condensation is the process where a gas transitions to the liquid phase when it is cooled. In an automotive air conditioning system, this is a crucial step to remove heat from the vehicle’s interior and create a cooling effect. When the refrigerant, usually R134a or R1234YF, flows through the evaporator, it absorbs heat from the ambient air inside the car. This heat absorption process cools the interior air. Since the refrigerant is exposed to relatively warm air at this point, it evaporates and transitions into the gaseous phase. This gas is then drawn into the compressor. In the compressor, the gaseous refrigerant is compressed, causing it to be under high pressure and significantly increase in temperature. This hot, compressed gas phase of the refrigerant exits the compressor and flows through the condenser.
Overheating: Overheating occurs when there is too little liquid refrigerant in the system, and the gas being compressed by the compressor becomes warmer than usual. This can have several negative effects on the performance of the air conditioning system and passenger comfort. If there is insufficient liquid to dissipate the heat generated by the compressor, the gas being compressed can reach an undesirably high temperature. This can result in reduced cooling capacity and even cause damage to the compressor and other components in the system. Moreover, overheating can impair the efficiency of the entire air conditioning system. The system will need to consume more energy to achieve the desired interior temperature, resulting in higher fuel consumption and potentially reducing the system’s lifespan.
Boiling Temperature: This is equivalent to the condensation temperature and depends on the pressure. The boiling temperature is the temperature at which a vaporous substance begins to condense and reverts to liquid form. In the case of an air conditioning system, the refrigerant, now in vapor form, is directed through the condenser section of the system. Here, heat is dissipated to the surrounding environment, cooling the refrigerant, causing it to condense into a liquid. This condensation process ensures that the heat absorbed in the evaporator is now released to the environment outside the air conditioning system.
Saturated Vapor: In an air conditioning system, saturated vapor occurs when both the vapor and liquid phases of the same refrigerant are present. In other words, the saturated vapor is the vapor that just begins to condense at a specific temperature and pressure.
Subcooling: Subcooling occurs when the liquid in the refrigerant is cooler than the boiling (condensation) temperature. This can cause various problems and negatively impact the air conditioning system’s performance. If the liquid is too cold, it can freeze as it flows through the evaporator. This can lead to ice formation on the evaporator and restricted airflow, resulting in a loss of cooling capacity and potential damage to the evaporator. Moreover, subcooling can decrease the overall efficiency of the system. It can cause the system to take longer to reach the desired temperature, making passengers less comfortable in the vehicle.
