Corrosion and rust:
The terms corrosion and rust are closely related. Rust is the result of corrosion. However, these two terms are often used interchangeably.
- Corrosion is a general term for the deterioration of material due to (chemical) reaction with its environment;
- Rust forms on the surface of iron or steel as a result of contact with water and oxygen. Rust refers to the oxidation of iron, which is also a form of corrosion.
When metals are exposed to the normal atmosphere (outdoor air), they form a chemical bond with oxygen. In this case, the metal starts to rust. Rust can be recognized by the reddish-brown layer that appears on the metal and develops when iron reacts with oxygen in the presence of moisture from the air or water. It is a mixture of iron oxide and iron hydroxide. Rust continues to develop and can completely corrode the metal. Over time, all the iron is converted into rust, causing it to lose its beneficial properties.
When iron is exposed to oxygen and water, over time, oxygen and iron bond at the atomic level. This creates a new compound: iron oxide. Water accelerates the whole process because it acts as a catalyst. The small water molecules penetrate the metal and form more acids, exposing the metal even more to oxidation. The rusting process is even faster in seawater due to the higher concentration of sodium chloride ions.
Rust spots can develop under car paint. This happens because the paint can also be somewhat porous, allowing moisture and oxygen to pass through. To keep the paint layer sealed, it’s a good idea to regularly wax the car. Especially in winter, with many damp conditions and road salt, this is a good way to prevent rust as much as possible.
When there are deep scratches or stone chips, the protective paint layer is missing at these spots, allowing rust to develop around them. It’s wise to touch up these spots with a paint stick or have them repainted as soon as possible.
In the image next to this, we see a VW Beetle with rusty body parts. This vehicle is classified as a “Rat rod.” Vehicles converted to Rat rod have intentionally worn and unpainted parts (in this case, rust) and discarded components.

In very few cases can rust be appreciated. Nevertheless, every vehicle will encounter it sooner or later. Besides the humid climate in the Netherlands, road salt in winter increases the risk of rust. Alongside cosmetic parts, parts of the chassis or bodywork can also be affected. In the long term, this can lead to an unsafe vehicle and failure during the APK (Dutch vehicle inspection).
Automakers try to make the materials in and around the car as rust-resistant as possible. Here are some of the rust-prevention treatments:
- galvanizing the bodywork;
- applying wax treatments;
- applying primer and base coats;
- applying high-quality paint layers.



Fighting rust:
Rust forms when iron has bonded with oxygen. It is incorrect to think that rust can be combated by preventing oxygen from reaching the iron. Steel is not a homogeneous substance but a mix of materials combined during recycling, such as old car wrecks. Other metals are also present in the steel. A small voltage difference exists between the crystals of two different materials, allowing a conductive liquid to create contact between the crystals. The resulting electrical current causes the less noble metal to dissolve. For example, if a current flows between iron and copper, the iron dissolves. The dissolved iron particles bond with oxygen. Even with the smallest amount of water in the bodywork layer, the process continues.
When rust is present under the paint, spraying over the rust is futile. The rust continues to corrode under the paint layer. Because the volume of rust is larger than that of steel, the paint layer will eventually crack, creating an opening where additional water can enter, accelerating the rusting process. To combat rust, steel is derusted by blasting the surface, followed by applying a rust-preventive primer layer as quickly as possible. Delaying this allows moisture from the air to bond with the iron particles.

Rust forms under the paint layer. Early-stage rust is therefore not always easy to recognize. In the four images below, a rocker panel of a VW Golf IV is shown where the rusty metal has been cut away. Before work began, there were only visible loose bitumen on the edge below the sill. The rocker panels showed no irregularities. When removing the bitumen, the brown edges became visible. The metal around it was already so corroded that it easily broke away. The photos show how rust can behave under bitumen.




Preventing rust:
In the first paragraph, “galvanizing” was mentioned in the list of measures to prevent rusting. Manufacturers apply a thin layer of zinc during the production of the car. This provides what is called cathodic protection. Since electrons will escape from zinc more readily, the zinc dissolves before the iron. Zinc more easily forms a bond with the oxygen in the air. The zinc is quite hard and forms a sealing layer. With deep scratches through the paint and zinc layer to the steel, the steel will hardly react. With sufficient zinc nearby, the electrical current will direct the right way. For this reason, zinc offers long-term protection against corrosion and rust.

Galvanized steel is not without disadvantages. Its increase reduces the amount of pure steel available. Manufacturers need zinc-free steel to make items like engine components. Zinc has the property of forming bubbles in the steel, resulting in weak spots.
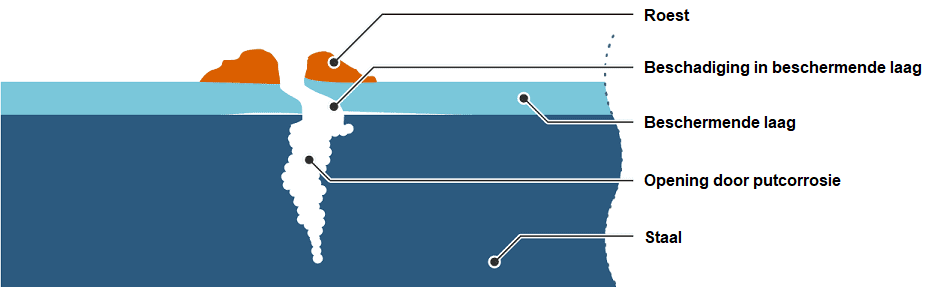
Pitting corrosion:
Pitting corrosion (English: pitting) is a localized attack on the material surface. Pitting corrosion occurs in materials that protect themselves against corrosion by an oxide layer and mainly develops where this oxide layer is damaged. Pitting corrodes quickly and deeply into the material. This is dangerous because the material may still appear intact, but the depth of the corrosion is not readily visible.
The image below shows the cross-section of a steel object with pitting corrosion. Only a small amount of rust is visible on top. The “pit” in the rust reveals the opening formed in the steel.
We can encounter pitting corrosion in, for example, brake lines. As the opening becomes deeper, there is a risk of brake fluid leaking out.

Contact corrosion:
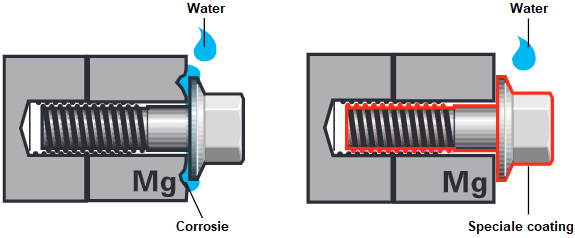
This occurs when two different metals come into close contact. A voltage difference is generated between these two metals, resulting in a reaction. We see this when, for example, a stainless steel screw is attached to a galvanized steel plate. The more noble metal promotes corrosion and starts to rust. This is called contact corrosion.
The following image shows two examples of a magnesium component, such as a gearbox, which is fastened with a bolt made of an iron alloy. When the contact surface is exposed to water, an electrical current is generated between the two metals, leading to contact corrosion. Simultaneously, the magnesium is degraded. Contact corrosion can be prevented by inhibiting the electrical current between the two metals by coating the bolt with a non-conductive, insulating layer.
The table below shows the different combinations of materials (such as engine parts or the gearbox housing) and how the choice of bolt or screw material affects contact corrosion.

